Research
Welcome to the Ralston Lab !
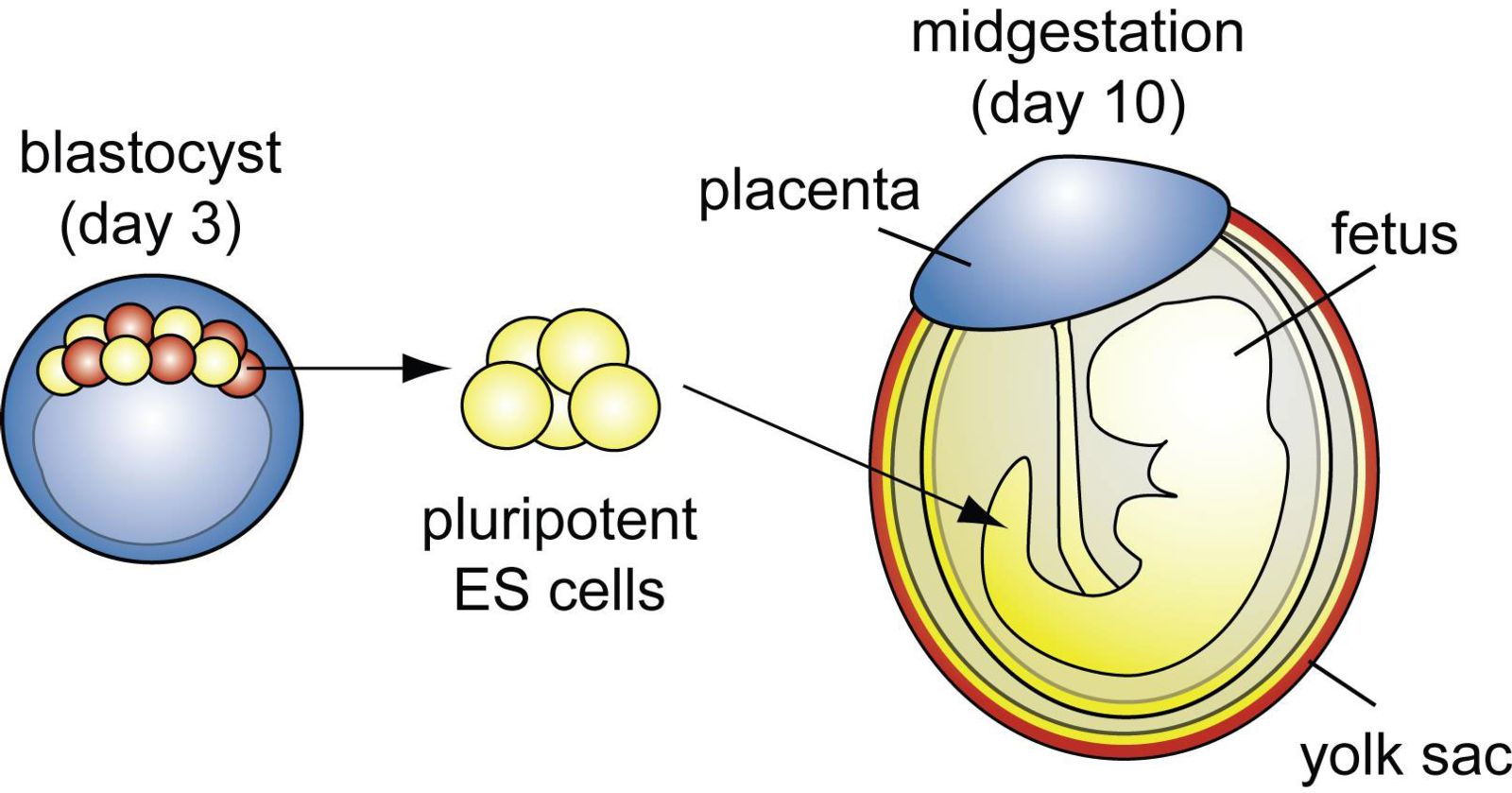
We study stem cells because they can be used to treat and study a variety of human diseases. Human stem cells can be derived from embryos, or they can be produced from patients using a revolutionary technique known as reprogramming. Remarkably, in some mammals, stem cells can be derived from the fetal membranes (such as the placenta) as well. Stem cells from the fetal membranes are important because they provide an exciting new approach to studying infertility, birth defects, cardiac and brain formation, and even Zika virus infection.
My lab uses stem cells approaches (differentiation, reprogramming), mouse genetic approaches (targeting, Cas9/CRISPR), genome-wide approaches (ChIP-seq, RNA-seq), single-cell approaches (flow cytometry, confocal microscopy), and proteomic approaches (western blot, mass spec) to discover how molecules instruct cell behavior. The product of our investigations is the discovery of how mutations and environmental insults cause human diseases and how to create and use stem cells to address these causes.

~ Header image of preimplantation mouse blastocyst courtesy of S. Blij. Footer image courtesy of MSU Sustainability/Be Spartan Green ~ Last updated 2023 with OMNI CMS by M. Halbisen.